LED Technology
Light-emitting diode

Red, green and blue LEDs of the 5mm type Type Passive, optoelectronic Working principle Electroluminescence Invented Nick Holonyak Jr. (1962) Electronic symbol Pin configuration Anode and Cathode
Pin configuration Anode and Cathode 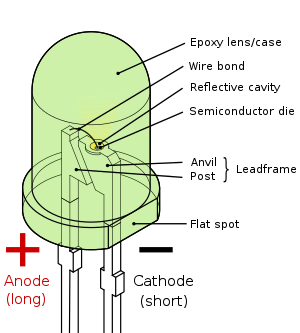

Parts of an LED


A light-emitting diode (LED) (pronounced /ˌɛl iː ˈdiː/, L-E-D[1]) is a semiconductor light source. LEDs are used as indicator lamps in many devices, and are increasingly used for lighting. Introduced as a practical electronic component in 1962,[2] early LEDs emitted low-intensity red light, but modern versions are available across the visible, ultraviolet and infrared wavelengths, with very high brightness.
When a light-emitting diode is forward biased (switched on), electrons are able to recombine with electron holes within the device, releasing energy in the form of photons. This effect is called electroluminescence and the color of the light (corresponding to the energy of the photon) is determined by the energy gap of the semiconductor. An LED is often small in area (less than 1 mm2), and integrated optical components may be used to shape its radiation pattern.[3] LEDs present many advantages over incandescent light sources including lower energy consumption, longer lifetime, improved robustness, smaller size, faster switching, and greater durability and reliability. LEDs powerful enough for room lighting are relatively expensive and require more precise current and heat management than compact fluorescent lamp sources of comparable output.
Light-emitting diodes are used in applications as diverse as replacements for aviation lighting, automotive lighting (particularly brake lamps, turn signals and indicators) as well as in traffic signals. The compact size, the possibility of narrow bandwidth, switching speed, and extreme reliability of LEDs has allowed new text and video displays and sensors to be developed, while their high switching rates are also useful in advanced communications technology. Infrared LEDs are also used in the remote control units of many commercial products including televisions, DVD players, and other domestic appliances.
Contents
Discoveries and early devices


Green electroluminescence from a point contact on a crystal of SiC recreates H. J. Round's original experiment from 1907.
Electroluminescence was discovered in 1907 by the British experimenter H. J. Round of Marconi Labs, using a crystal of silicon carbide and a cat's-whisker detector.[4][5] Russian Oleg Vladimirovich Losev independently reported on the creation of an LED in 1927.[6][7] His research was distributed in Russian, German and British scientific journals, but no practical use was made of the discovery for several decades.[8][9] Rubin Braunstein of the Radio Corporation of America reported on infrared emission from gallium arsenide (GaAs) and other semiconductor alloys in 1955.[10] Braunstein observed infrared emission generated by simple diode structures using gallium antimonide (GaSb), GaAs, indium phosphide (InP), and silicon-germanium (SiGe) alloys at room temperature and at 77 kelvin.
In 1961, American experimenters Robert Biard and Gary Pittman working at Texas Instruments,[11] found that GaAs emitted infrared radiation when electric current was applied and received the patent for the infrared LED.
The first practical visible-spectrum (red) LED was developed in 1962 by Nick Holonyak Jr., while working at General Electric Company.[2] Holonyak is seen as the "father of the light-emitting diode".[12] M. George Craford,[13] a former graduate student of Holonyak, invented the first yellow LED and improved the brightness of red and red-orange LEDs by a factor of ten in 1972.[14] In 1976, T.P. Pearsall created the first high-brightness, high efficiency LEDs for optical fiber telecommunications by inventing new semiconductor materials specifically adapted to optical fiber transmission wavelengths.[15]
Until 1968, visible and infrared LEDs were extremely costly, on the order of US $200 per unit, and so had little practical use.[16] The Monsanto Company was the first organization to mass-produce visible LEDs, using gallium arsenide phosphide in 1968 to produce red LEDs suitable for indicators.[16] Hewlett Packard (HP) introduced LEDs in 1968, initially using GaAsP supplied by Monsanto. The technology proved to have major uses for alphanumeric displays and was integrated into HP's early handheld calculators. In the 1970s commercially successful LED devices at under five cents each were produced by Fairchild Optoelectronics. These devices employed compound semiconductor chips fabricated with the planar process invented by Dr. Jean Hoerni at Fairchild Semiconductor.[17] The combination of planar processing for chip fabrication and innovative packaging methods enabled the team at Fairchild led by optoelectronics pioneer Thomas Brandt to achieve the needed cost reductions. These methods continue to be used by LED producers.[18]
Practical use
The first commercial LEDs were commonly used as replacements for incandescent and neon indicator lamps, and in seven-segment displays,[19] first in expensive equipment such as laboratory and electronics test equipment, then later in such appliances as TVs, radios, telephones, calculators, and even watches (see list of signal uses). These red LEDs were bright enough only for use as indicators, as the light output was not enough to illuminate an area. Readouts in calculators were so small that plastic lenses were built over each digit to make them legible. Later, other colors grew widely available and also appeared in appliances and equipment. As LED materials technology grew more advanced, light output rose, while maintaining efficiency and reliability at acceptable levels. The invention and development of the high power white light LED led to use for illumination[20][21] (see list of illumination applications). Most LEDs were made in the very common 5 mm T1¾ and 3 mm T1 packages, but with rising power output, it has grown increasingly necessary to shed excess heat to maintain reliability,[22] so more complex packages have been adapted for efficient heat dissipation. Packages for state-of-the-art high power LEDs bear little resemblance to early LEDs.


Illustration of Haitz's Law. Light output per LED as a function of production year, note the logarithmic scale on the vertical axis.
Continuing development
The first high-brightness blue LED was demonstrated by Shuji Nakamura of Nichia Corporation and was based on InGaN borrowing on critical developments in GaN nucleation on sapphire substrates and the demonstration of p-type doping of GaN which were developed by Isamu Akasaki and H. Amano in Nagoya. In 1995, Alberto Barbieri at the Cardiff University Laboratory (GB) investigated the efficiency and reliability of high-brightness LEDs and demonstrated a very impressive result by using a transparent contact made of indium tin oxide (ITO) on (AlGaInP/GaAs) LED. The existence of blue LEDs and high efficiency LEDs quickly led to the development of the first white LED, which employed a Y3Al5O12:Ce, or "YAG", phosphor coating to mix yellow (down-converted) light with blue to produce light that appears white. Nakamura was awarded the 2006 Millennium Technology Prize for his invention.[23]
The development of LED technology has caused their efficiency and light output to rise exponentially, with a doubling occurring about every 36 months since the 1960s, in a way similar to Moore's law. The advances are generally attributed to the parallel development of other semiconductor technologies and advances in optics and material science. This trend is normally called Haitz's Law after Dr. Roland Haitz. [24]
In February 2008, 300 lumens of visible light per watt luminous efficacy (not per electrical watt) and warm-light emission was achieved by using nanocrystals.[25]
In 2009, a process for growing gallium nitride (GaN) LEDs on silicon has been reported. Epitaxy costs could be reduced by up to 90% using six-inch silicon wafers instead of two-inch sapphire wafers.[26]
Technology


The inner workings of an LED
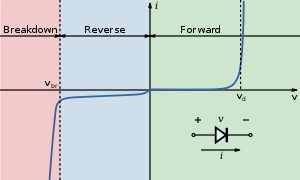

I-V diagram for a diode. An LED will begin to emit light when the on-voltage is exceeded. Typical on voltages are 2–3 volts
Physics
Like a normal diode, the LED consists of a chip of semiconducting material doped with impurities to create a p-n junction. As in other diodes, current flows easily from the p-side, or anode, to the n-side, or cathode, but not in the reverse direction. Charge-carriers—electrons and holes—flow into the junction from electrodes with different voltages. When an electron meets a hole, it falls into a lower energy level, and releases energy in the form of a photon.
The wavelength of the light emitted, and thus its color, depends on the band gap energy of the materials forming the p-n junction. In silicon or germanium diodes, the electrons and holes recombine by a non-radiative transition which produces no optical emission, because these are indirect band gap materials. The materials used for the LED have a direct band gap with energies corresponding to near-infrared, visible or near-ultraviolet light.
LED development began with infrared and red devices made with gallium arsenide. Advances in materials science have enabled making devices with ever-shorter wavelengths, emitting light in a variety of colors.
LEDs are usually built on an n-type substrate, with an electrode attached to the p-type layer deposited on its surface. P-type substrates, while less common, occur as well. Many commercial LEDs, especially GaN/InGaN, also use sapphire substrate.
Most materials used for LED production have very high refractive indices. This means that much light will be reflected back into the material at the material/air surface interface. Thus, light extraction in LEDs is an important aspect of LED production, subject to much research and development.
Refractive Index


Idealized example of light emission cones in a semiconductor, for a single point-source emission zone. The left illustration is for a fully translucent wafer, while the right illustration shows the half-cones formed when the bottom layer is fully opaque. The light is actually emitted equally in all directions from the point-source, so the areas between the cones shows the large amount of trapped light energy that is wasted as heat.[27]


The light emission cones of a real LED wafer are far more complex than a single point-source light emission. Typically the light emission zone is a 2D plane between the wafers. Across this 2D plane, there is effectively a separate set of emission cones for every atom.
Drawing the billions of overlapping cones is impossible, so this is a simplified diagram showing the extents of all the emission cones combined. The larger side cones are clipped to show the interior features and reduce image complexity; they would extend to the opposite edges of the 2D emission plane.
Bare uncoated semiconductors such as silicon exhibit a very high refractive index relative to open air, which prevents passage of photons at sharp angles relative to the air-contacting surface of the semiconductor. This property affects both the light-emission efficiency of LEDs as well as the light-absorption efficiency of photovoltaic cells. The refractive index of silicon is 4.24, while air is 1.00002926[28]
Generally a flat-surfaced uncoated LED semiconductor chip will only emit light perpendicular to the semiconductor's surface, and a few degrees to the side, in a cone shape referred to as the light cone, cone of light,[29] or the escape cone.[27] The maximum angle of incidence is referred to as the critical angle. When this angle is exceeded photons no longer penetrate the semiconductor, but are instead reflected both internally inside the semiconductor crystal, and externally off the surface of the crystal as if it were a mirror.[27]
Internal reflections can escape through other crystalline faces, if the incidence angle is low enough and the crystal is sufficiently transparent to not re-absorb the photon emission. But for a simple square LED with 90-degree angled surfaces on all sides, the faces all act as equal angle mirrors. In this case the light can not escape and is lost as waste heat in the crystal.[27]
A convoluted chip surface with angled facets similar to a jewel or fresnel lens can increase light output by allowing light to be emitted perpendicular to the chip surface while far to the sides of the photon emission point.[30]
The ideal shape of a semiconductor with maximum light output would be a microsphere with the photon emission occurring at the exact center, with electrodes penetrating to the center to contact at the emission point. All light rays emanating from the center would be perpendicular to the entire surface of the sphere, resulting in no internal reflections. A hemispherical semiconductor would also work, with the flat back-surface serving as a mirror to back-scattered photons.[31]
Transition coatings
Many LED semiconductor chips are potted in clear or colored molded plastic shells. The plastic shell has three purposes:
Efficiency and operational parameters
Typical indicator LEDs are designed to operate with no more than 30–60 milliwatts [mW] of electrical power. Around 1999, Philips Lumileds introduced power LEDs capable of continuous use at one watt [W]. These LEDs used much larger semiconductor die sizes to handle the large power inputs. Also, the semiconductor dies were mounted onto metal slugs to allow for heat removal from the LED die.
One of the key advantages of LED-based lighting is its high efficiency, as measured by its light output per unit power input. White LEDs quickly matched and overtook the efficiency of standard incandescent lighting systems. In 2002, Lumileds made five-watt LEDs available with a luminous efficacy of 18–22 lumens per watt [lm/W]. For comparison, a conventional 60–100 W incandescent lightbulb emits around 15 lm/W, and standard fluorescent lights emit up to 100 lm/W. A recurring problem is that efficiency falls sharply with rising current. This effect is known as droop and effectively limits the light output of a given LED, raising heating more than light output for higher current.[33][34][35]
In September 2003, a new type of blue LED was demonstrated by the company Cree Inc. to provide 24 mW at 20 milliamperes [mA]. This produced a commercially packaged white light giving 65 lm/W at 20 mA, becoming the brightest white LED commercially available at the time, and more than four times as efficient as standard incandescents. In 2006, they demonstrated a prototype with a record white LED luminous efficacy of 131 lm/W at 20 mA. Also, Seoul Semiconductor plans for 135 lm/W by 2007 and 145 lm/W by 2008,[36] which would be nearing an order of magnitude improvement over standard incandescents and better than even standard fluorescents. Nichia Corporation has developed a white LED with luminous efficacy of 150 lm/W at a forward current of 20 mA.[37]
Practical general lighting needs high-power LEDs, of one watt or more. Typical operating currents for such devices begin at 350 mA.
Note that these efficiencies are for the LED chip only, held at low temperature in a lab. Lighting works at higher temperature and with drive circuit losses, so efficiencies are much lower. United States Department of Energy (DOE) testing of commercial LED lamps designed to replace incandescent lamps or CFLs showed that average efficacy was still about 46 lm/W in 2009 (tested performance ranged from 17 lm/W to 79 lm/W).[38]
Cree issued a press release on February 3, 2010 about a laboratory prototype LED achieving 208 lumens per watt at room temperature. The correlated color temperature was reported to be 4579 K.[39]
Lifetime and failure
Main article: List of LED failure modes
Solid state devices such as LEDs are subject to very limited wear and tear if operated at low currents and at low temperatures. Many of the LEDs made in the 1970s and 1980s are still in service today. Typical lifetimes quoted are 25,000 to 100,000 hours but heat and current settings can extend or shorten this time significantly. [40]
The most common symptom of LED (and diode laser) failure is the gradual lowering of light output and loss of efficiency. Sudden failures, although rare, can occur as well. Early red LEDs were notable for their short lifetime. With the development of high-power LEDs the devices are subjected to higher junction temperatures and higher current densities than traditional devices. This causes stress on the material and may cause early light output degradation. To quantitatively classify lifetime in a standardized manner it has been suggested to use the terms L75 and L50 which is the time it will take a given LED to reach 75% and 50% light output respectively.[41]
Like other lighting devices, LED performance is temperature dependent. Most manufacturers’ published ratings of LEDs are for an operating temperature of 25°C. LEDs used outdoors, such as traffic signals or in-pavement signal lights, and that are utilized in climates where the temperature within the luminaire gets very hot, could result in low signal intensities or even failure.[42]
LED light output actually rises at colder temperatures (leveling off depending on type at around −30C[citation needed]). Consequently, LED technology may be a good replacement in uses such as supermarket freezer lighting[43][44][45] and will last longer than other technologies. Because LEDs emit less heat than incandescent bulbs, they are an energy-efficient technology for uses such as freezers. However, because they emit little heat, ice and snow may build up on the LED luminaire in colder climates.[42] This lack of waste heat generation has been observed to cause sometimes significant problems with street traffic signals and airport runway lighting in snow-prone areas, although some research has been done to try to develop heat sink technologies to transfer heat to other areas of the luminaire.[46]
Ultraviolet and blue LEDs


Blue LEDs.
Blue LEDs are based on the wide band gap semiconductors GaN (gallium nitride) and InGaN (indium gallium nitride). They can be added to existing red and green LEDs to produce the impression of white light, though white LEDs today rarely use this principle.
The first blue LEDs were made in 1971 by Jacques Pankove (inventor of the gallium nitride LED) at RCA Laboratories.[53] These devices had too little light output to be of much practical use. In the late 1980s, key breakthroughs in GaN epitaxial growth and p-type doping[54] ushered in the modern era of GaN-based optoelectronic devices. Building upon this foundation, in 1993 high brightness blue LEDs were demonstrated.[55]
By the late 1990s, blue LEDs had become widely available. They have an active region consisting of one or more InGaN quantum wells sandwiched between thicker layers of GaN, called cladding layers. By varying the relative InN-GaN fraction in the InGaN quantum wells, the light emission can be varied from violet to amber. AlGaN aluminium gallium nitride of varying AlN fraction can be used to manufacture the cladding and quantum well layers for ultraviolet LEDs, but these devices have not yet reached the level of efficiency and technological maturity of the InGaN-GaN blue/green devices. If the active quantum well layers are GaN, instead of alloyed InGaN or AlGaN, the device will emit near-ultraviolet light with wavelengths around 350–370 nm. Green LEDs manufactured from the InGaN-GaN system are far more efficient and brighter than green LEDs produced with non-nitride material systems.
With nitrides containing aluminium, most often AlGaN and AlGaInN, even shorter wavelengths are achievable. Ultraviolet LEDs in a range of wavelengths are becoming available on the market. Near-UV emitters at wavelengths around 375–395 nm are already cheap and often encountered, for example, as black light lamp replacements for inspection of anti-counterfeiting UV watermarks in some documents and paper currencies. Shorter wavelength diodes, while substantially more expensive, are commercially available for wavelengths down to 247 nm.[56] As the photosensitivity of microorganisms approximately matches the absorption spectrum of DNA, with a peak at about 260 nm, UV LED emitting at 250–270 nm are to be expected in prospective disinfection and sterilization devices. Recent research has shown that commercially available UVA LEDs (365 nm) are already effective disinfection and sterilization devices.[57]
Deep-UV wavelengths were obtained in laboratories using aluminium nitride (210 nm),[51] boron nitride (215 nm)[49][50] and diamond (235 nm).[48]
White light
There are two primary ways of producing high intensity white-light using LEDs. One is to use individual LEDs that emit three primary colors[58]—red, green, and blue—and then mix all the colors to form white light. The other is to use a phosphor material to convert monochromatic light from a blue or UV LED to broad-spectrum white light, much in the same way a fluorescent light bulb works.
Due to metamerism, it is possible to have quite different spectra that appear white.
RGB systems


Combined spectral curves for blue, yellow-green, and high brightness red solid-state semiconductor LEDs. FWHM spectral bandwidth is approximately 24–27 nm for all three colors.
White light can be formed by mixing differently colored light, the most common method is to use red, green and blue (RGB). Hence the method is called multi-colored white LEDs (sometimes referred to as RGB LEDs). Because its mechanism is involved with electro-optical devices to control the blending and diffusion of different colors, this means is little used to produce white lighting. Nevertheless, this method is particularly interesting in many uses because of the flexibility of mixing different colors,[59] and, in principle, this mechanism also has higher quantum efficiency in producing white light.
There are several types of multi-colored white LEDs: di-, tri-, and tetrachromatic white LEDs. Several key factors that play among these different methods, include color stability, color rendering capability, and luminous efficacy. Often higher efficiency will mean lower color rendering, presenting a trade off between the luminous efficiency and color rendering. For example, the dichromatic white LEDs have the best luminous efficacy (120 lm/W), but the lowest color rendering capability. Conversely, although tetrachromatic white LEDs have excellent color rendering capability, they often have poor luminous efficiency. Trichromatic white LEDs are in between, having both good luminous efficacy (>70 lm/W) and fair color rendering capability.
Multi-color LEDs offer not merely another means to form white light, but a new means to form light of different colors. Most perceivable colors can be formed by mixing different amounts of three primary colors. This allows precise dynamic color control. As more effort is devoted to investigating this method, multi-color LEDs should have profound influence on the fundamental method which we use to produce and control light color. However, before this type of LED can play a role on the market, several technical problems need solving. These include that this type of LED's emission power decays exponentially with rising temperature,[60] resulting in a substantial change in color stability. Such problems inhibit and may preclude industrial use. Thus, many new package designs aimed at solving this problem have been proposed and their results are now being reproduced by researchers and scientists.
Phosphor-based LEDs


Spectrum of a “white” LED clearly showing blue light which is directly emitted by the GaN-based LED (peak at about 465 nm) and the more broadband Stokes-shifted light emitted by the Ce3+:YAG phosphor which emits at roughly 500–700 nm.
This method involves coating an LED of one color (mostly blue LED made of InGaN) with phosphor of different colors to form white light; the resultant LEDs are called phosphor-based white LEDs.[61] A fraction of the blue light undergoes the Stokes shift being transformed from shorter wavelengths to longer. Depending on the color of the original LED, phosphors of different colors can be employed. If several phosphor layers of distinct colors are applied, the emitted spectrum is broadened, effectively raising the color rendering index (CRI) value of a given LED.[62]
Phosphor based LEDs have a lower efficiency than normal LEDs due to the heat loss from the Stokes shift and also other phosphor-related degradation issues. However, the phosphor method is still the most popular method for making high intensity white LEDs. The design and production of a light source or light fixture using a monochrome emitter with phosphor conversion is simpler and cheaper than a complex RGB system, and the majority of high intensity white LEDs presently on the market are manufactured using phosphor light conversion.
The greatest barrier to high efficiency is the seemingly unavoidable Stokes energy loss. However, much effort is being spent on optimizing these devices to higher light output and higher operation temperatures. For instance, the efficiency can be raised by adapting better package design or by using a more suitable type of phosphor. Philips Lumileds' patented conformal coating process addresses the issue of varying phosphor thickness, giving the white LEDs a more homogeneous white light.[63] With development ongoing, the efficiency of phosphor based LEDs generally rises with each new product announcement.
The phosphor based white LEDs encapsulate InGaN blue LEDs inside phosphor coated epoxy. A common yellow phosphor material is cerium-doped yttrium aluminium garnet (Ce3+:YAG).
White LEDs can also be made by coating near ultraviolet (NUV) emitting LEDs with a mixture of high efficiency europium-based red and blue emitting phosphors plus green emitting copper and aluminium doped zinc sulfide (ZnS:Cu, Al). This is a method analogous to the way fluorescent lamps work. This method is less efficient than the blue LED with YAG:Ce phosphor, as the Stokes shift is larger, so more energy is converted to heat, but yields light with better spectral characteristics, which render color better. Due to the higher radiative output of the ultraviolet LEDs than of the blue ones, both methods offer comparable brightness. A concern is that UV light may leak from a malfunctioning light source and cause harm to human eyes or skin.
Other white LEDs
Another method used to produce experimental white light LEDs used no phosphors at all and was based on homoepitaxially grown zinc selenide (ZnSe) on a ZnSe substrate which simultaneously emitted blue light from its active region and yellow light from the substrate.[64]
Organic light-emitting diodes (OLEDs)
Main article: Organic light-emitting diode
If the emitting layer material of the LED is an organic compound, it is known as an organic light emitting diode (OLED). To function as a semiconductor, the organic emitting material needs conjugated pi bonds. [65] The emitting material can be a small organic molecule in a crystalline phase, or a polymer. Polymer materials can be flexible; such LEDs are known as PLEDs or FLEDs.
Compared with regular LEDs, OLEDs are lighter, and polymer LEDs can have the added benefit of being flexible. Some possible future uses of OLEDs may be:
As of 2008, OLEDs operated at substantially lower efficiency than inorganic (crystalline) LEDs.[67]
Quantum dot LEDs (experimental)
A new method developed by Michael Bowers, a graduate student at Vanderbilt University in Nashville, involves coating a blue LED with quantum dots that glow white in response to the blue light from the LED. This method emits a warm, yellowish-white light similar to that made by incandescent bulbs.[68]
Quantum dots are semiconductor nanocrystals that possess unique optical properties.[69] Their emission color can be tuned from the visible throughout the infrared spectrum. This allows quantum dot LEDs to create almost any color on the CIE diagram. This provides more color options and better color rendering than white LEDs. Quantum dot LEDs are available in the same package types as traditional phosphor based LEDs.
In September 2009 Nanoco Group announced that it has signed a joint development agreement with a major Japanese electronics company under which it will design and develop quantum dots for use in light emitting diodes (LEDs) in liquid crystal display (LCD) televisions.[70]
Types


LEDs are produced in a variety of shapes and sizes. The 5 mm cylindrical package (red, fifth from the left) is the most common, estimated at 80% of world production.[citation needed] The color of the plastic lens is often the same as the actual color of light emitted, but not always. For instance, purple plastic is often used for infrared LEDs, and most blue devices have clear housings. There are also LEDs in SMT packages, such as those found on blinkies and on cell phone keypads (not shown).
The main types of LEDs are miniature, high power devices and custom designs such as alphanumeric or multi-color.
Miniature


Different sized LEDs. 8 mm, 5 mm and 3 mm, with a wooden match-stick for scale.
Main article: Miniature light-emitting diode
These are mostly single-die LEDs used as indicators, and they come in various-sizes from 2 mm to 8 mm, through-hole and surface mount packages. They are usually simple in design, not requiring any separate cooling body.[71] Typical current ratings ranges from around 1 mA to above 20 mA. The small scale sets a natural upper boundary on power consumption due to heat caused by the high current density and need for heat sinking.
Mid-range
Medium power LEDs are often through-hole mounted and used when an output of a few lumen is needed. They sometimes have the diode mounted to four leads (two cathode leads, two anode leads) for better heat conduction and carry an integrated lens. An example of this is the Superflux package, from Philips Lumileds. These LEDs are most commonly used in light panels, emergency lighting and automotive tail-lights. Due to the larger amount of metal in the LED, they are able to handle higher currents (around 100 mA). The higher current allows for the higher light output required for tail-lights and emergency lighting.
High power
See also: Solid-state lighting and LED lamp


High-power light emiting diodes (Luxeon, Lumileds)
High power LEDs (HPLED) can be driven at currents from hundreds of mA to more than an ampere, compared with the tens of mA for other LEDs. Some can emit over a thousand lumens.[72][73] Since overheating is destructive, the HPLEDs must be mounted on a heat sink to allow for heat dissipation. If the heat from a HPLED is not removed, the device will fail in seconds. One HPLED can often replace an incandescent bulb in a torch, or be set in an array to form a powerful LED lamp.
Some well-known HPLEDs in this category are the Lumileds Rebel Led, Osram Opto Semiconductors Golden Dragon and Cree X-lamp. As of September 2009 some HPLEDs manufactured by Cree Inc. now exceed 105 lm/W [74] (e.g. the XLamp XP-G LED chip emitting Cool White light) and are being sold in lamps intended to replace incandescent, halogen, and even fluorescent lights, as LEDs grow more cost competitive.
LEDs have been developed by Seoul Semiconductor that can operate on AC power without the need for a DC converter. For each half cycle, part of the LED emits light and part is dark, and this is reversed during the next half cycle. The efficacy of this type of HPLED is typically 40 lm/W.[75] A large number of LED elements in series may be able to operate directly from line voltage. In 2009 Seoul Semiconductor released a high DC voltage capable of being driven from AC power with a simple controlling circuit. The low power dissipation of these LEDs affords them more flexibility than the original AC LED design.[76]
Application-specific variations


Calculator LED display, 1970s.
Power sources
Main article: LED power sources
The current/voltage characteristic of an LED is similar to other diodes, in that the current is dependent exponentially on the voltage (see Shockley diode equation). This means that a small change in voltage can cause a large change in current. If the maximum voltage rating is exceeded by a small amount, the current rating may be exceeded by a large amount, potentially damaging or destroying the LED. The typical solution is to use constant current power supplies, or driving the LED at a voltage much below the maximum rating. Since most common power sources (batteries, mains) are not constant current sources, most LED fixtures must include a power converter. However, the I/V curve of nitride-based LEDs is quite steep above the knee and gives an If of a few milliamperes at a Vf of 3 V, making it possible to power a nitride-based LED from a 3 V battery such as a coin cell without the need for a current limiting resistor.
Electrical polarity
Main article: Electrical polarity of LEDs
As with all diodes, current flows easily from p-type to n-type material.[77] However, no current flows and no light is emitted if a small voltage is applied in the reverse direction. If the reverse voltage grows large enough to exceed the breakdown voltage, a large current flows and the LED may be damaged. If the reverse current is sufficiently limited to avoid damage, the reverse-conducting LED is a useful noise diode.
Safety
The vast majority of devices containing LEDs are "safe under all conditions of normal use", and so are classified as "Class 1 LED product"/"LED Klasse 1". At present, only a few LEDs—extremely bright LEDs that also have a tightly focused viewing angle of 8° or less—could, in theory, cause temporary blindness, and so are classified as "Class 2".[78] In general, laser safety regulations—and the "Class 1", "Class 2", etc. system—also apply to LEDs.[79]
Advantages


LED lighting in the aircraft cabin of an Airbus A320 Enhanced.


A large LED display behind a disc jockey.


LED destination displays on buses, one with a colored route number.


LED digital display that can display 4 digits along with points.


Traffic light using LED


Western Australia Police car using LED


Printhead of an Oki LED printer


LED daytime running lights of Audi A4


LED panel light source used in an experiment on plant growth. The findings of such experiments may be used to grow food in space on long duration missions.
LED uses fall into four major categories:
The low energy consumption, low maintenance and small size of modern LEDs has led to uses as status indicators and displays on a variety of equipment and installations. Large area LED displays are used as stadium displays and as dynamic decorative displays. Thin, lightweight message displays are used at airports and railway stations, and as destination displays for trains, buses, trams, and ferries.
One-color light is well suited for traffic lights and signals, exit signs, emergency vehicle lighting, ships' navigation lights or lanterns (chromacity and luminance standards being set under the Convention on the International Regulations for Preventing Collisions at Sea 1972, Annex I and the CIE) and LED-based Christmas lights. In cold climates, LED traffic lights may remain snow covered.[96] Red or yellow LEDs are used in indicator and alphanumeric displays in environments where night vision must be retained: aircraft cockpits, submarine and ship bridges, astronomy observatories, and in the field, e.g. night time animal watching and military field use.
Because of their long life and fast switching times, LEDs have been used in brake lights for cars high-mounted brake lights, trucks, and buses, and in turn signals for some time, but many vehicles now use LEDs for their rear light clusters. The use in brakes improves safety, due to a great reduction in the time needed to light fully, or faster rise time, up to 0.5 second faster than an incandescent bulb. This gives drivers behind more time to react. It is reported that at normal highway speeds, this equals one car length equivalent in increased time to react. In a dual intensity circuit (i.e., rear markers and brakes) if the LEDs are not pulsed at a fast enough frequency, they can create a phantom array, where ghost images of the LED will appear if the eyes quickly scan across the array. White LED headlamps are starting to be used. Using LEDs has styling advantages because LEDs can form much thinner lights than incandescent lamps with parabolic reflectors.
Due to the relative cheapness of low output LEDs, they are also used in many temporary uses such as glowsticks, throwies, and the photonic textile Lumalive. Artists have also used LEDs for LED art.
Weather/all-hazards radio receivers with Specific Area Message Encoding (SAME) have three LEDs: red for warnings, orange for watches, and yellow for advisories & statements whenever issued.
Lighting
Main article: LED lamp
With the development of high efficiency and high power LEDs it has grown possible to use LEDs in lighting and illumination. Replacement light bulbs have been made, as well as dedicated fixtures and LED lamps. LEDs are used as street lights and in other architectural lighting where color changing is used. The mechanical robustness and long lifetime is used in automotive lighting on cars, motorcycles and on bicycle lights.
LED street lights are employed on poles and in parking garages. In 2007, the Italian village Torraca was the first place to convert its entire illumination system to LEDs.[97]
LEDs are used in aviation lighting. Airbus has used LED lighting in their Airbus A320 Enhanced since 2007, and Boeing plans its use in the 787. LEDs are also being used now in airport and heliport lighting. LED airport fixtures currently include medium intensity runway lights, runway centerline lights and obstruction lighting.
LEDs are also suitable for backlighting for LCD televisions and lightweight laptop displays and light source for DLP projectors (See LED TV). RGB LEDs raise the color gamut by as much as 45%. Screens for TV and computer displays can be made thinner using LEDs for backlighting.[98]
LEDs are used increasingly commonly in aquarium lights. Particularly for reef aquariums, LED lights provide an efficient light source with less heat output to help maintain optimal aquarium temperatures. LED-based aquarium fixtures also have the advantage of being manually adjustable to emit a specific color-spectrum for ideal coloration of corals, fish, and invertebrates while optimizing photosynthetically active radiation (PAR) which raises growth and sustainability of photosynthetic life such as corals, anemones, clams, and macroalgae. These fixtures can be electronically programmed to simulate various lighting conditions throughout the day, reflecting phases of the sun and moon for a dynamic reef experience. LED fixtures typically cost up to five times as much as similarly rated fluorescent or high-intensity discharge lighting designed for reef aquariums and are not as high output to date.
The lack of IR/heat radiation makes LEDs ideal for stage lights using banks of RGB LEDs that can easily change color and decrease heating from traditional stage lighting, as well as medical lighting where IR-radiation can be harmful.
LEDs are small, durable and need little power, so they are used in hand held devices such as flashlights. LED strobe lights or camera flashes operate at a safe, low voltage, instead of the 250+ volts commonly found in xenon flashlamp-based lighting. This is especially useful in cameras on mobile phones, where space is at a premium and bulky voltage-raising circuitry is undesirable. LEDs are used for infrared illumination in night vision uses including security cameras. A ring of LEDs around a video camera, aimed forward into a retroreflective background, allows chroma keying in video productions.
LEDs are used for decorative lighting as well. Uses include but are not limited to indoor/outdoor decor, limousines, cargo trailers, conversion vans, cruise ships, RVs, boats, automobiles, and utility trucks. Decorative LED lighting can also come in the form of lighted company signage and step and aisle lighting in theaters and auditoriums.
Smart lighting
Light can be used to transmit broadband data, which is already implemented in IrDA standards using infrared LEDs. Because LEDs can cycle on and off millions of times per second, they can be wireless transmitters and access points for data transport.[99] Lasers can also be modulated in this manner.
Sustainable lighting
Efficient lighting is needed for sustainable architecture. A 13 watt LED lamp emits 450 to 650 lumens.[100] which is equivalent to a standard 40 watt incandescent bulb. [101] A standard 40 W incandescent bulb has an expected lifespan of 1,000 hours while an LED can continue to operate with reduced efficiency for more than 50,000 hours, 50 times longer than the incandescent bulb.
Environmentally friendly options
One kilowatt-hour of electricity will cause 1.34 pounds (610 g) of CO2 emission.[102] Assuming the average light bulb is on for 10 hours a day, one 40-watt incandescent bulb will cause 196 pounds (89 kg) of CO2 emission per year. The 13-watt LED equivalent will only cause 63 pounds (29 kg) of CO2 over the same time span. A building’s carbon footprint from lighting can be reduced by 68% by exchanging all incandescent bulbs for new LEDs in warm climates. In cold climates, the energy saving may be lower, since more heating is needed to compensate for the lower temperature.
LEDs are also non-toxic unlike the more popular energy efficient bulb option: the compact fluorescent a.k.a. CFL which contains traces of harmful mercury. While the amount of mercury in a CFL is small, introducing less into the environment is preferable.
Economically sustainable
LED light bulbs could be a cost-effective option for lighting a home or office space because of their very long lifetimes. Consumer use of LEDs as a replacement for conventional lighting system is currently hampered by the high cost and low efficiency of available products. 2009 DOE testing results showed an average efficacy of 35 lm/W, below that of typical CFLs, and as low as 9 lm/W, worse than standard incandescents.[100] The high initial cost of the commercial LED bulb is due to the expensive sapphire substrate which is key to the production process. The sapphire apparatus must be coupled with a mirror-like collector to reflect light that would otherwise be wasted.
In 2008, a materials science research team at Purdue University succeeded in making LED bulbs with a substitute for the sapphire components.[103] The team used metal-coated silicon wafers with a built-in reflective layer of zirconium nitride to lessen the overall production cost of the LED. They predict that within a few years, LEDs produced with their revolutionary new method will be competitively priced with CFLs. The less expensive LED would not only be the best energy saver, but also a low cost bulb.
Non-visual applications
Light has many other uses besides for seeing. LEDs are used for some of these. The uses fall in three groups: Communication, sensors and light matter interaction.
The light from LEDs can be modulated very quickly so they are used extensively in optical fiber and Free Space Optics communications. This include remote controls, such as for TVs and VCRs, where infrared LEDs are often used. Opto-isolators use an LED combined with a photodiode or phototransistor to provide a signal path with electrical isolation between two circuits. This is especially useful in medical equipment where the signals from a low voltage sensor circuit (usually battery powered) in contact with a living organism must be electrically isolated from any possible electrical failure in a recording or monitoring device operating at potentially dangerous voltages. An optoisolator also allows information to be transferred between circuits not sharing a common ground potential.
Many sensor systems rely on light as the signal source. LEDs are often ideal as a light source due to the requirements of the sensors. LEDs are used as movement sensors, for example in optical computer mice. The Nintendo Wii's sensor bar uses infrared LEDs. In pulse oximeters for measuring oxygen saturation. Some flatbed scanners use arrays of RGB LEDs rather than the typical cold-cathode fluorescent lamp as the light source. Having independent control of three illuminated colors allows the scanner to calibrate itself for more accurate color balance, and there is no need for warm-up. Further, its sensors only need be monochromatic, since at any one time the page being scanned is only lit by one color of light. Touch sensing: Since LEDs can also be used as photodiodes, they can be used for both photo emission and detection. This could be used in for example a touch-sensing screen that register reflected light from a finger or stylus.[104]
Many materials and biological systems are sensitive to, or dependent on light. Grow lights use LEDs to increase photosynthesis in plants[105] and bacteria and viruses can be removed from water and other substances using UV LEDs for sterilization.[57] Other uses are as UV curing devices for some ink and coating methods, and in LED printers.
Plant growers are interested in LEDs because they are more energy efficient, emit less heat (can damage plants close to hot lamps), and can provide the optimum light frequency for plant growth and bloom periods compared to currently used grow lights: HPS (high pressure sodium), MH (metal halide) or CFL/low-energy. However, LEDs have not replaced these grow lights due to higher price. As mass production and LED kits develop, the LED products will become cheaper.
LEDs have also been used as a medium quality voltage reference in electronic circuits. The forward voltage drop (e.g., about 1.7 V for a normal red LED) can be used instead of a Zener diode in low-voltage regulators. Red LEDs have the flattest I/V curve above the knee. Nitride-based LEDs have a fairly steep I/V curve and are useless for this purpose. Although LED forward voltage is far more current-dependent than a good Zener, Zener diodes are not widely available below voltages of about 3 V.
Light sources for machine vision systems
Machine vision systems often require bright and homogeneous illumination, so features of interest are easier to process. LEDs are often used for this purpose, and this is likely to remain one of their major uses until price drops low enough to make signaling and illumination uses more widespread. Barcode scanners are the most common example of machine vision, and many low cost ones use red LEDs instead of lasers. Optical computer mice are also another example of LEDs in machine vision, as it is used to provide an even light source on the surface for the miniature camera within the mouse. LEDs constitute a nearly ideal light source for machine vision systems for several reasons:
The size of the illuminated field is usually comparatively small and machine vision systems are often quite expensive, so the cost of the light source is usually a minor concern. However, it might not be easy to replace a broken light source placed within complex machinery, and here the long service life of LEDs is a benefit.
LED elements tend to be small and can be placed with high density over flat or even shaped substrates (PCBs etc.) so that bright and homogeneous sources can be designed which direct light from tightly controlled directions on inspected parts. This can often be obtained with small, low cost lenses and diffusers, helping to achieve high light densities with control over lighting levels and homogeneity. LED sources can be shaped in several configurations (spot lights for reflective illumination; ring lights for coaxial illumination; back lights for contour illumination; linear assemblies; flat, large format panels; dome sources for diffused, omnidirectional illumination).
LEDs can be easily strobed (in the microsecond range and below) and synchronized with imaging. High power LEDs are available allowing well lit images even with very short light pulses. This is often used to obtain crisp and sharp “still” images of quickly moving parts.
LEDs come in several different colors and wavelengths, allowing easy use of the best color for each need, where different color may provide better visibility of features of interest. Having a precisely known spectrum allows tightly matched filters to be used to separate informative bandwidth or to reduce disturbing effects of ambient light. LEDs usually operate at comparatively low working temperatures, simplifying heat management and dissipation. This allows using plastic lenses, filters, and diffusers. Waterproof units can also easily be designed, allowing use in harsh or wet environments (food, beverage, oil industries).
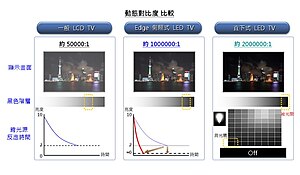

Comparison of LCD, edge lit LED and LED TVs
A LED-backlight LCD television is an LCD TV that uses LED backlighting[1][2] rather than the cold cathode fluorescent lights (CCFLs) used in traditional LCD televisions. It is not a true LED display but is often called "LED TV" by some manufacturers.[3] The use of LED backlighting has a dramatic impact, resulting in a thinner panel, less power consumption, and a brighter display with better contrast levels. It also generates less heat than an ordinary LCD TV.
The LEDs can come in three forms: dynamic RGB LEDs which are positioned behind the panel, white Edge-LEDs positioned around the rim of the screen which use a special diffusion panel to spread the light evenly behind the screen (the most common) and full-array which are arranged behind the screen but they are incapable of dimming or brightening individually.[4][5]
LED backlighting techniques
RGB dynamic LEDs
This method of backlighting allows dimming to occur in locally specific areas of darkness on the screen. This can show truer blacks, whites and PRs[clarification needed] at much higher dynamic contrast ratios, at the cost of less detail in small bright objects on a dark background, such as star fields.[6]
Edge-LEDs
This method of backlighting allows for LED-backlit TVs to become extremely thin. The light is diffused across the screen by a special panel which produces a uniform color range across the screen.
Full Array LEDs
Many brands use LED backlighting technology and may offer a range of benefits over CCFL LCD TVs such as reduced energy consumption, better contrast and brightness, greater colour range, more rapid response to changes in scene and a capacity to provide the means to render an image more accurately.[7]
Differences between LED-backlit and CCFL-backlit LCD displays
LED-backlit LCD TVs differ from conventional CCFL-backlit LCD TVs in the following:
TV manufacturers can use an LED backlight instead of the standard Cold Cathode Fluorescent Lamps (LCD-CCFL) used in most LCD televisions. It is important to distinguish this method of simply backlighting a conventional LCD panel, from a hypothetical true LED display, or an Organic light-emitting diode (OLED) display. LCD-based televisions described as 'LED TVs' are vastly different from self-illuminating OLED, OEL or AMOLED display technologies. In terms of the use of the term 'LED TV' in the UK, the ASA (Advertising Standards Authority) has made it clear in prior correspondence that it does not object to the use of the term, but does require it to be clarified in any advertising. There are several methods of backlighting an LCD panel using LEDs including the use of either White or RGB (Red, Green and Blue) LED arrays positioned behind the panel; and Edge-LED lighting, which uses white LEDs arranged around the inside frame of the TV along with a special light diffusion panel designed to spread the light evenly behind the LCD panel.
An LED backlight offers several general benefits over regular CCFL backlight TVs, typically higher brightness. Compared to regular CCFL backlighting, there may also be benefits to color gamut. However advancements in CCFL technology mean wide color gamuts and lower power consumption are also possible. The principal barrier to wide use of LED backlighting on LCD televisions is cost.
The variations of LED backlighting do offer different benefits. The first commercial LED backlit LCD TV was the Sony Qualia 005 (introduced in 2004). This featured RGB LED arrays to offer a color gamut around twice that of a conventional CCFL LCD television (the combined light output from red, green and blue LEDs produces a more pure white light than is possible with a single white light LED). RGB LED technology continues to be used on selected Sony BRAVIA LCD models, with the addition of 'local dimming' which enables excellent on-screen contrast through selectively turning off the LEDs behind dark parts of a picture frame.
Edge LED lighting was also first introduced by Sony (September 2008) on the 40 inch BRAVIA KLV-40ZX1M (referred to as the ZX1 in Europe). The principal benefit of Edge-LED lighting for LCD televisions is the ability to build thinner housings (the BRAVIA KLV-40ZX1M is as thin as 9.9mm). Samsung has also introduced a range of Edge-LED lit LCD televisions with extremely thin housings.
LED-backlit LCD TVs are considered a more sustainable choice, with a longer life and better energy efficiency than plasmas and conventional LCD TVs.[10] Unlike CCFL backlights, LEDs also use no mercury in their manufacture. However, other elements such as gallium and arsenic are used in the manufacture of the LED emitters themselves, meaning there is some debate over whether they are a significantly better long term solution to the problem of TV disposal.
Because LEDs are able to be switched on and off more quickly than CCFL displays and can offer a higher light output, it is theoretically possible to offer very high contrast ratios. They can produce deep blacks (LEDs off) and a high brightness (LEDs on), however care should be taken with measurements made from pure black and pure white outputs, as technologies like Edge-LED lighting do not allow these outputs to be reproduced simultaneously on-screen.
In September 2009 Nanoco Group announced that it has signed a joint development agreement with a major Japanese electronics company under which it will design and develop quantum dots for LED Backlights in LCD televisions.[11] Quantum dots are valued for displays, because they emit light in very specific gaussian distributions. This can result in a display that more accurately renders the colors than the human eye can perceive. Quantum dots also require very little power since they are not color filtered. In September 2010, LG Electronics revealed their new product which claimed as the world's slimmest full LED 3D TV at the IFA consumer electronics trade show in Berlin.[12]
Light-emitting diode

Red, green and blue LEDs of the 5mm type Type Passive, optoelectronic Working principle Electroluminescence Invented Nick Holonyak Jr. (1962) Electronic symbol
 Pin configuration Anode and Cathode
Pin configuration Anode and Cathode 

Parts of an LED


A light-emitting diode (LED) (pronounced /ˌɛl iː ˈdiː/, L-E-D[1]) is a semiconductor light source. LEDs are used as indicator lamps in many devices, and are increasingly used for lighting. Introduced as a practical electronic component in 1962,[2] early LEDs emitted low-intensity red light, but modern versions are available across the visible, ultraviolet and infrared wavelengths, with very high brightness.
When a light-emitting diode is forward biased (switched on), electrons are able to recombine with electron holes within the device, releasing energy in the form of photons. This effect is called electroluminescence and the color of the light (corresponding to the energy of the photon) is determined by the energy gap of the semiconductor. An LED is often small in area (less than 1 mm2), and integrated optical components may be used to shape its radiation pattern.[3] LEDs present many advantages over incandescent light sources including lower energy consumption, longer lifetime, improved robustness, smaller size, faster switching, and greater durability and reliability. LEDs powerful enough for room lighting are relatively expensive and require more precise current and heat management than compact fluorescent lamp sources of comparable output.
Light-emitting diodes are used in applications as diverse as replacements for aviation lighting, automotive lighting (particularly brake lamps, turn signals and indicators) as well as in traffic signals. The compact size, the possibility of narrow bandwidth, switching speed, and extreme reliability of LEDs has allowed new text and video displays and sensors to be developed, while their high switching rates are also useful in advanced communications technology. Infrared LEDs are also used in the remote control units of many commercial products including televisions, DVD players, and other domestic appliances.
Contents
- 1 History
- 1.1 Discoveries and early devices
- 1.2 Practical use
- 1.3 Continuing development
- 2 Technology
- 2.1 Physics
- 2.2 Refractive Index
- 2.2.1 Transition coatings
- 2.3 Efficiency and operational parameters
- 2.4 Lifetime and failure
- 3 Colors and materials
- 3.1 Ultraviolet and blue LEDs
- 3.2 White light
- 3.2.1 RGB systems
- 3.2.2 Phosphor-based LEDs
- 3.2.3 Other white LEDs
- 3.3 Organic light-emitting diodes (OLEDs)
- 3.4 Quantum dot LEDs (experimental)
- 4 Types
- 4.1 Miniature
- 4.2 Mid-range
- 4.3 High power
- 4.4 Application-specific variations
- 5 Considerations for use
- 5.1 Power sources
- 5.2 Electrical polarity
- 5.3 Safety
- 5.4 Advantages
- 5.5 Disadvantages
- 6 Applications
- 6.1 Indicators and signs
- 6.2 Lighting
- 6.3 Smart lighting
- 6.4 Sustainable lighting
- 6.4.1 Environmentally friendly options
- 6.4.2 Economically sustainable
- 6.5 Non-visual applications
- 6.6 Light sources for machine vision systems
- 7 See also
- 8 References
- 8.1 Notes
- 8.2 Further reading
- 9 External links
Discoveries and early devices


Green electroluminescence from a point contact on a crystal of SiC recreates H. J. Round's original experiment from 1907.
Electroluminescence was discovered in 1907 by the British experimenter H. J. Round of Marconi Labs, using a crystal of silicon carbide and a cat's-whisker detector.[4][5] Russian Oleg Vladimirovich Losev independently reported on the creation of an LED in 1927.[6][7] His research was distributed in Russian, German and British scientific journals, but no practical use was made of the discovery for several decades.[8][9] Rubin Braunstein of the Radio Corporation of America reported on infrared emission from gallium arsenide (GaAs) and other semiconductor alloys in 1955.[10] Braunstein observed infrared emission generated by simple diode structures using gallium antimonide (GaSb), GaAs, indium phosphide (InP), and silicon-germanium (SiGe) alloys at room temperature and at 77 kelvin.
In 1961, American experimenters Robert Biard and Gary Pittman working at Texas Instruments,[11] found that GaAs emitted infrared radiation when electric current was applied and received the patent for the infrared LED.
The first practical visible-spectrum (red) LED was developed in 1962 by Nick Holonyak Jr., while working at General Electric Company.[2] Holonyak is seen as the "father of the light-emitting diode".[12] M. George Craford,[13] a former graduate student of Holonyak, invented the first yellow LED and improved the brightness of red and red-orange LEDs by a factor of ten in 1972.[14] In 1976, T.P. Pearsall created the first high-brightness, high efficiency LEDs for optical fiber telecommunications by inventing new semiconductor materials specifically adapted to optical fiber transmission wavelengths.[15]
Until 1968, visible and infrared LEDs were extremely costly, on the order of US $200 per unit, and so had little practical use.[16] The Monsanto Company was the first organization to mass-produce visible LEDs, using gallium arsenide phosphide in 1968 to produce red LEDs suitable for indicators.[16] Hewlett Packard (HP) introduced LEDs in 1968, initially using GaAsP supplied by Monsanto. The technology proved to have major uses for alphanumeric displays and was integrated into HP's early handheld calculators. In the 1970s commercially successful LED devices at under five cents each were produced by Fairchild Optoelectronics. These devices employed compound semiconductor chips fabricated with the planar process invented by Dr. Jean Hoerni at Fairchild Semiconductor.[17] The combination of planar processing for chip fabrication and innovative packaging methods enabled the team at Fairchild led by optoelectronics pioneer Thomas Brandt to achieve the needed cost reductions. These methods continue to be used by LED producers.[18]
Practical use
The first commercial LEDs were commonly used as replacements for incandescent and neon indicator lamps, and in seven-segment displays,[19] first in expensive equipment such as laboratory and electronics test equipment, then later in such appliances as TVs, radios, telephones, calculators, and even watches (see list of signal uses). These red LEDs were bright enough only for use as indicators, as the light output was not enough to illuminate an area. Readouts in calculators were so small that plastic lenses were built over each digit to make them legible. Later, other colors grew widely available and also appeared in appliances and equipment. As LED materials technology grew more advanced, light output rose, while maintaining efficiency and reliability at acceptable levels. The invention and development of the high power white light LED led to use for illumination[20][21] (see list of illumination applications). Most LEDs were made in the very common 5 mm T1¾ and 3 mm T1 packages, but with rising power output, it has grown increasingly necessary to shed excess heat to maintain reliability,[22] so more complex packages have been adapted for efficient heat dissipation. Packages for state-of-the-art high power LEDs bear little resemblance to early LEDs.


Illustration of Haitz's Law. Light output per LED as a function of production year, note the logarithmic scale on the vertical axis.
Continuing development
The first high-brightness blue LED was demonstrated by Shuji Nakamura of Nichia Corporation and was based on InGaN borrowing on critical developments in GaN nucleation on sapphire substrates and the demonstration of p-type doping of GaN which were developed by Isamu Akasaki and H. Amano in Nagoya. In 1995, Alberto Barbieri at the Cardiff University Laboratory (GB) investigated the efficiency and reliability of high-brightness LEDs and demonstrated a very impressive result by using a transparent contact made of indium tin oxide (ITO) on (AlGaInP/GaAs) LED. The existence of blue LEDs and high efficiency LEDs quickly led to the development of the first white LED, which employed a Y3Al5O12:Ce, or "YAG", phosphor coating to mix yellow (down-converted) light with blue to produce light that appears white. Nakamura was awarded the 2006 Millennium Technology Prize for his invention.[23]
The development of LED technology has caused their efficiency and light output to rise exponentially, with a doubling occurring about every 36 months since the 1960s, in a way similar to Moore's law. The advances are generally attributed to the parallel development of other semiconductor technologies and advances in optics and material science. This trend is normally called Haitz's Law after Dr. Roland Haitz. [24]
In February 2008, 300 lumens of visible light per watt luminous efficacy (not per electrical watt) and warm-light emission was achieved by using nanocrystals.[25]
In 2009, a process for growing gallium nitride (GaN) LEDs on silicon has been reported. Epitaxy costs could be reduced by up to 90% using six-inch silicon wafers instead of two-inch sapphire wafers.[26]
Technology


The inner workings of an LED


I-V diagram for a diode. An LED will begin to emit light when the on-voltage is exceeded. Typical on voltages are 2–3 volts
Physics
Like a normal diode, the LED consists of a chip of semiconducting material doped with impurities to create a p-n junction. As in other diodes, current flows easily from the p-side, or anode, to the n-side, or cathode, but not in the reverse direction. Charge-carriers—electrons and holes—flow into the junction from electrodes with different voltages. When an electron meets a hole, it falls into a lower energy level, and releases energy in the form of a photon.
The wavelength of the light emitted, and thus its color, depends on the band gap energy of the materials forming the p-n junction. In silicon or germanium diodes, the electrons and holes recombine by a non-radiative transition which produces no optical emission, because these are indirect band gap materials. The materials used for the LED have a direct band gap with energies corresponding to near-infrared, visible or near-ultraviolet light.
LED development began with infrared and red devices made with gallium arsenide. Advances in materials science have enabled making devices with ever-shorter wavelengths, emitting light in a variety of colors.
LEDs are usually built on an n-type substrate, with an electrode attached to the p-type layer deposited on its surface. P-type substrates, while less common, occur as well. Many commercial LEDs, especially GaN/InGaN, also use sapphire substrate.
Most materials used for LED production have very high refractive indices. This means that much light will be reflected back into the material at the material/air surface interface. Thus, light extraction in LEDs is an important aspect of LED production, subject to much research and development.
Refractive Index


Idealized example of light emission cones in a semiconductor, for a single point-source emission zone. The left illustration is for a fully translucent wafer, while the right illustration shows the half-cones formed when the bottom layer is fully opaque. The light is actually emitted equally in all directions from the point-source, so the areas between the cones shows the large amount of trapped light energy that is wasted as heat.[27]


The light emission cones of a real LED wafer are far more complex than a single point-source light emission. Typically the light emission zone is a 2D plane between the wafers. Across this 2D plane, there is effectively a separate set of emission cones for every atom.
Drawing the billions of overlapping cones is impossible, so this is a simplified diagram showing the extents of all the emission cones combined. The larger side cones are clipped to show the interior features and reduce image complexity; they would extend to the opposite edges of the 2D emission plane.
Bare uncoated semiconductors such as silicon exhibit a very high refractive index relative to open air, which prevents passage of photons at sharp angles relative to the air-contacting surface of the semiconductor. This property affects both the light-emission efficiency of LEDs as well as the light-absorption efficiency of photovoltaic cells. The refractive index of silicon is 4.24, while air is 1.00002926[28]
Generally a flat-surfaced uncoated LED semiconductor chip will only emit light perpendicular to the semiconductor's surface, and a few degrees to the side, in a cone shape referred to as the light cone, cone of light,[29] or the escape cone.[27] The maximum angle of incidence is referred to as the critical angle. When this angle is exceeded photons no longer penetrate the semiconductor, but are instead reflected both internally inside the semiconductor crystal, and externally off the surface of the crystal as if it were a mirror.[27]
Internal reflections can escape through other crystalline faces, if the incidence angle is low enough and the crystal is sufficiently transparent to not re-absorb the photon emission. But for a simple square LED with 90-degree angled surfaces on all sides, the faces all act as equal angle mirrors. In this case the light can not escape and is lost as waste heat in the crystal.[27]
A convoluted chip surface with angled facets similar to a jewel or fresnel lens can increase light output by allowing light to be emitted perpendicular to the chip surface while far to the sides of the photon emission point.[30]
The ideal shape of a semiconductor with maximum light output would be a microsphere with the photon emission occurring at the exact center, with electrodes penetrating to the center to contact at the emission point. All light rays emanating from the center would be perpendicular to the entire surface of the sphere, resulting in no internal reflections. A hemispherical semiconductor would also work, with the flat back-surface serving as a mirror to back-scattered photons.[31]
Transition coatings
Many LED semiconductor chips are potted in clear or colored molded plastic shells. The plastic shell has three purposes:
- Mounting the semiconductor chip in devices is easier to accomplish.
- The tiny fragile electrical wiring is physically supported and protected from damage
- The plastic acts as a refractive intermediary between the relatively high-index semiconductor and low-index open air.[32]
Efficiency and operational parameters
Typical indicator LEDs are designed to operate with no more than 30–60 milliwatts [mW] of electrical power. Around 1999, Philips Lumileds introduced power LEDs capable of continuous use at one watt [W]. These LEDs used much larger semiconductor die sizes to handle the large power inputs. Also, the semiconductor dies were mounted onto metal slugs to allow for heat removal from the LED die.
One of the key advantages of LED-based lighting is its high efficiency, as measured by its light output per unit power input. White LEDs quickly matched and overtook the efficiency of standard incandescent lighting systems. In 2002, Lumileds made five-watt LEDs available with a luminous efficacy of 18–22 lumens per watt [lm/W]. For comparison, a conventional 60–100 W incandescent lightbulb emits around 15 lm/W, and standard fluorescent lights emit up to 100 lm/W. A recurring problem is that efficiency falls sharply with rising current. This effect is known as droop and effectively limits the light output of a given LED, raising heating more than light output for higher current.[33][34][35]
In September 2003, a new type of blue LED was demonstrated by the company Cree Inc. to provide 24 mW at 20 milliamperes [mA]. This produced a commercially packaged white light giving 65 lm/W at 20 mA, becoming the brightest white LED commercially available at the time, and more than four times as efficient as standard incandescents. In 2006, they demonstrated a prototype with a record white LED luminous efficacy of 131 lm/W at 20 mA. Also, Seoul Semiconductor plans for 135 lm/W by 2007 and 145 lm/W by 2008,[36] which would be nearing an order of magnitude improvement over standard incandescents and better than even standard fluorescents. Nichia Corporation has developed a white LED with luminous efficacy of 150 lm/W at a forward current of 20 mA.[37]
Practical general lighting needs high-power LEDs, of one watt or more. Typical operating currents for such devices begin at 350 mA.
Note that these efficiencies are for the LED chip only, held at low temperature in a lab. Lighting works at higher temperature and with drive circuit losses, so efficiencies are much lower. United States Department of Energy (DOE) testing of commercial LED lamps designed to replace incandescent lamps or CFLs showed that average efficacy was still about 46 lm/W in 2009 (tested performance ranged from 17 lm/W to 79 lm/W).[38]
Cree issued a press release on February 3, 2010 about a laboratory prototype LED achieving 208 lumens per watt at room temperature. The correlated color temperature was reported to be 4579 K.[39]
Lifetime and failure
Main article: List of LED failure modes
Solid state devices such as LEDs are subject to very limited wear and tear if operated at low currents and at low temperatures. Many of the LEDs made in the 1970s and 1980s are still in service today. Typical lifetimes quoted are 25,000 to 100,000 hours but heat and current settings can extend or shorten this time significantly. [40]
The most common symptom of LED (and diode laser) failure is the gradual lowering of light output and loss of efficiency. Sudden failures, although rare, can occur as well. Early red LEDs were notable for their short lifetime. With the development of high-power LEDs the devices are subjected to higher junction temperatures and higher current densities than traditional devices. This causes stress on the material and may cause early light output degradation. To quantitatively classify lifetime in a standardized manner it has been suggested to use the terms L75 and L50 which is the time it will take a given LED to reach 75% and 50% light output respectively.[41]
Like other lighting devices, LED performance is temperature dependent. Most manufacturers’ published ratings of LEDs are for an operating temperature of 25°C. LEDs used outdoors, such as traffic signals or in-pavement signal lights, and that are utilized in climates where the temperature within the luminaire gets very hot, could result in low signal intensities or even failure.[42]
LED light output actually rises at colder temperatures (leveling off depending on type at around −30C[citation needed]). Consequently, LED technology may be a good replacement in uses such as supermarket freezer lighting[43][44][45] and will last longer than other technologies. Because LEDs emit less heat than incandescent bulbs, they are an energy-efficient technology for uses such as freezers. However, because they emit little heat, ice and snow may build up on the LED luminaire in colder climates.[42] This lack of waste heat generation has been observed to cause sometimes significant problems with street traffic signals and airport runway lighting in snow-prone areas, although some research has been done to try to develop heat sink technologies to transfer heat to other areas of the luminaire.[46]
Ultraviolet and blue LEDs


Blue LEDs.
Blue LEDs are based on the wide band gap semiconductors GaN (gallium nitride) and InGaN (indium gallium nitride). They can be added to existing red and green LEDs to produce the impression of white light, though white LEDs today rarely use this principle.
The first blue LEDs were made in 1971 by Jacques Pankove (inventor of the gallium nitride LED) at RCA Laboratories.[53] These devices had too little light output to be of much practical use. In the late 1980s, key breakthroughs in GaN epitaxial growth and p-type doping[54] ushered in the modern era of GaN-based optoelectronic devices. Building upon this foundation, in 1993 high brightness blue LEDs were demonstrated.[55]
By the late 1990s, blue LEDs had become widely available. They have an active region consisting of one or more InGaN quantum wells sandwiched between thicker layers of GaN, called cladding layers. By varying the relative InN-GaN fraction in the InGaN quantum wells, the light emission can be varied from violet to amber. AlGaN aluminium gallium nitride of varying AlN fraction can be used to manufacture the cladding and quantum well layers for ultraviolet LEDs, but these devices have not yet reached the level of efficiency and technological maturity of the InGaN-GaN blue/green devices. If the active quantum well layers are GaN, instead of alloyed InGaN or AlGaN, the device will emit near-ultraviolet light with wavelengths around 350–370 nm. Green LEDs manufactured from the InGaN-GaN system are far more efficient and brighter than green LEDs produced with non-nitride material systems.
With nitrides containing aluminium, most often AlGaN and AlGaInN, even shorter wavelengths are achievable. Ultraviolet LEDs in a range of wavelengths are becoming available on the market. Near-UV emitters at wavelengths around 375–395 nm are already cheap and often encountered, for example, as black light lamp replacements for inspection of anti-counterfeiting UV watermarks in some documents and paper currencies. Shorter wavelength diodes, while substantially more expensive, are commercially available for wavelengths down to 247 nm.[56] As the photosensitivity of microorganisms approximately matches the absorption spectrum of DNA, with a peak at about 260 nm, UV LED emitting at 250–270 nm are to be expected in prospective disinfection and sterilization devices. Recent research has shown that commercially available UVA LEDs (365 nm) are already effective disinfection and sterilization devices.[57]
Deep-UV wavelengths were obtained in laboratories using aluminium nitride (210 nm),[51] boron nitride (215 nm)[49][50] and diamond (235 nm).[48]
White light
There are two primary ways of producing high intensity white-light using LEDs. One is to use individual LEDs that emit three primary colors[58]—red, green, and blue—and then mix all the colors to form white light. The other is to use a phosphor material to convert monochromatic light from a blue or UV LED to broad-spectrum white light, much in the same way a fluorescent light bulb works.
Due to metamerism, it is possible to have quite different spectra that appear white.
RGB systems


Combined spectral curves for blue, yellow-green, and high brightness red solid-state semiconductor LEDs. FWHM spectral bandwidth is approximately 24–27 nm for all three colors.
White light can be formed by mixing differently colored light, the most common method is to use red, green and blue (RGB). Hence the method is called multi-colored white LEDs (sometimes referred to as RGB LEDs). Because its mechanism is involved with electro-optical devices to control the blending and diffusion of different colors, this means is little used to produce white lighting. Nevertheless, this method is particularly interesting in many uses because of the flexibility of mixing different colors,[59] and, in principle, this mechanism also has higher quantum efficiency in producing white light.
There are several types of multi-colored white LEDs: di-, tri-, and tetrachromatic white LEDs. Several key factors that play among these different methods, include color stability, color rendering capability, and luminous efficacy. Often higher efficiency will mean lower color rendering, presenting a trade off between the luminous efficiency and color rendering. For example, the dichromatic white LEDs have the best luminous efficacy (120 lm/W), but the lowest color rendering capability. Conversely, although tetrachromatic white LEDs have excellent color rendering capability, they often have poor luminous efficiency. Trichromatic white LEDs are in between, having both good luminous efficacy (>70 lm/W) and fair color rendering capability.
Multi-color LEDs offer not merely another means to form white light, but a new means to form light of different colors. Most perceivable colors can be formed by mixing different amounts of three primary colors. This allows precise dynamic color control. As more effort is devoted to investigating this method, multi-color LEDs should have profound influence on the fundamental method which we use to produce and control light color. However, before this type of LED can play a role on the market, several technical problems need solving. These include that this type of LED's emission power decays exponentially with rising temperature,[60] resulting in a substantial change in color stability. Such problems inhibit and may preclude industrial use. Thus, many new package designs aimed at solving this problem have been proposed and their results are now being reproduced by researchers and scientists.
Phosphor-based LEDs


Spectrum of a “white” LED clearly showing blue light which is directly emitted by the GaN-based LED (peak at about 465 nm) and the more broadband Stokes-shifted light emitted by the Ce3+:YAG phosphor which emits at roughly 500–700 nm.
This method involves coating an LED of one color (mostly blue LED made of InGaN) with phosphor of different colors to form white light; the resultant LEDs are called phosphor-based white LEDs.[61] A fraction of the blue light undergoes the Stokes shift being transformed from shorter wavelengths to longer. Depending on the color of the original LED, phosphors of different colors can be employed. If several phosphor layers of distinct colors are applied, the emitted spectrum is broadened, effectively raising the color rendering index (CRI) value of a given LED.[62]
Phosphor based LEDs have a lower efficiency than normal LEDs due to the heat loss from the Stokes shift and also other phosphor-related degradation issues. However, the phosphor method is still the most popular method for making high intensity white LEDs. The design and production of a light source or light fixture using a monochrome emitter with phosphor conversion is simpler and cheaper than a complex RGB system, and the majority of high intensity white LEDs presently on the market are manufactured using phosphor light conversion.
The greatest barrier to high efficiency is the seemingly unavoidable Stokes energy loss. However, much effort is being spent on optimizing these devices to higher light output and higher operation temperatures. For instance, the efficiency can be raised by adapting better package design or by using a more suitable type of phosphor. Philips Lumileds' patented conformal coating process addresses the issue of varying phosphor thickness, giving the white LEDs a more homogeneous white light.[63] With development ongoing, the efficiency of phosphor based LEDs generally rises with each new product announcement.
The phosphor based white LEDs encapsulate InGaN blue LEDs inside phosphor coated epoxy. A common yellow phosphor material is cerium-doped yttrium aluminium garnet (Ce3+:YAG).
White LEDs can also be made by coating near ultraviolet (NUV) emitting LEDs with a mixture of high efficiency europium-based red and blue emitting phosphors plus green emitting copper and aluminium doped zinc sulfide (ZnS:Cu, Al). This is a method analogous to the way fluorescent lamps work. This method is less efficient than the blue LED with YAG:Ce phosphor, as the Stokes shift is larger, so more energy is converted to heat, but yields light with better spectral characteristics, which render color better. Due to the higher radiative output of the ultraviolet LEDs than of the blue ones, both methods offer comparable brightness. A concern is that UV light may leak from a malfunctioning light source and cause harm to human eyes or skin.
Other white LEDs
Another method used to produce experimental white light LEDs used no phosphors at all and was based on homoepitaxially grown zinc selenide (ZnSe) on a ZnSe substrate which simultaneously emitted blue light from its active region and yellow light from the substrate.[64]
Organic light-emitting diodes (OLEDs)
Main article: Organic light-emitting diode
If the emitting layer material of the LED is an organic compound, it is known as an organic light emitting diode (OLED). To function as a semiconductor, the organic emitting material needs conjugated pi bonds. [65] The emitting material can be a small organic molecule in a crystalline phase, or a polymer. Polymer materials can be flexible; such LEDs are known as PLEDs or FLEDs.
Compared with regular LEDs, OLEDs are lighter, and polymer LEDs can have the added benefit of being flexible. Some possible future uses of OLEDs may be:
- Low cost, flexible displays
- Light sources
- Wall decorations
- Luminous cloth
As of 2008, OLEDs operated at substantially lower efficiency than inorganic (crystalline) LEDs.[67]
Quantum dot LEDs (experimental)
A new method developed by Michael Bowers, a graduate student at Vanderbilt University in Nashville, involves coating a blue LED with quantum dots that glow white in response to the blue light from the LED. This method emits a warm, yellowish-white light similar to that made by incandescent bulbs.[68]
Quantum dots are semiconductor nanocrystals that possess unique optical properties.[69] Their emission color can be tuned from the visible throughout the infrared spectrum. This allows quantum dot LEDs to create almost any color on the CIE diagram. This provides more color options and better color rendering than white LEDs. Quantum dot LEDs are available in the same package types as traditional phosphor based LEDs.
In September 2009 Nanoco Group announced that it has signed a joint development agreement with a major Japanese electronics company under which it will design and develop quantum dots for use in light emitting diodes (LEDs) in liquid crystal display (LCD) televisions.[70]
Types


LEDs are produced in a variety of shapes and sizes. The 5 mm cylindrical package (red, fifth from the left) is the most common, estimated at 80% of world production.[citation needed] The color of the plastic lens is often the same as the actual color of light emitted, but not always. For instance, purple plastic is often used for infrared LEDs, and most blue devices have clear housings. There are also LEDs in SMT packages, such as those found on blinkies and on cell phone keypads (not shown).
The main types of LEDs are miniature, high power devices and custom designs such as alphanumeric or multi-color.
Miniature


Different sized LEDs. 8 mm, 5 mm and 3 mm, with a wooden match-stick for scale.
Main article: Miniature light-emitting diode
These are mostly single-die LEDs used as indicators, and they come in various-sizes from 2 mm to 8 mm, through-hole and surface mount packages. They are usually simple in design, not requiring any separate cooling body.[71] Typical current ratings ranges from around 1 mA to above 20 mA. The small scale sets a natural upper boundary on power consumption due to heat caused by the high current density and need for heat sinking.
Mid-range
Medium power LEDs are often through-hole mounted and used when an output of a few lumen is needed. They sometimes have the diode mounted to four leads (two cathode leads, two anode leads) for better heat conduction and carry an integrated lens. An example of this is the Superflux package, from Philips Lumileds. These LEDs are most commonly used in light panels, emergency lighting and automotive tail-lights. Due to the larger amount of metal in the LED, they are able to handle higher currents (around 100 mA). The higher current allows for the higher light output required for tail-lights and emergency lighting.
High power
See also: Solid-state lighting and LED lamp


High-power light emiting diodes (Luxeon, Lumileds)
High power LEDs (HPLED) can be driven at currents from hundreds of mA to more than an ampere, compared with the tens of mA for other LEDs. Some can emit over a thousand lumens.[72][73] Since overheating is destructive, the HPLEDs must be mounted on a heat sink to allow for heat dissipation. If the heat from a HPLED is not removed, the device will fail in seconds. One HPLED can often replace an incandescent bulb in a torch, or be set in an array to form a powerful LED lamp.
Some well-known HPLEDs in this category are the Lumileds Rebel Led, Osram Opto Semiconductors Golden Dragon and Cree X-lamp. As of September 2009 some HPLEDs manufactured by Cree Inc. now exceed 105 lm/W [74] (e.g. the XLamp XP-G LED chip emitting Cool White light) and are being sold in lamps intended to replace incandescent, halogen, and even fluorescent lights, as LEDs grow more cost competitive.
LEDs have been developed by Seoul Semiconductor that can operate on AC power without the need for a DC converter. For each half cycle, part of the LED emits light and part is dark, and this is reversed during the next half cycle. The efficacy of this type of HPLED is typically 40 lm/W.[75] A large number of LED elements in series may be able to operate directly from line voltage. In 2009 Seoul Semiconductor released a high DC voltage capable of being driven from AC power with a simple controlling circuit. The low power dissipation of these LEDs affords them more flexibility than the original AC LED design.[76]
Application-specific variations
- Flashing LEDs are used as attention seeking indicators without requiring external electronics. Flashing LEDs resemble standard LEDs but they contain an integrated multivibrator circuit which causes the LED to flash with a typical period of one second. In diffused lens LEDs this is visible as a small black dot. Most flashing LEDs emit light of one color, but more sophisticated devices can flash between multiple colors and even fade through a color sequence using RGB color mixing.


Calculator LED display, 1970s.
- Bi-color LEDs are actually two different LEDs in one case. They consist of two dies connected to the same two leads antiparallel to each other. Current flow in one direction emits one color, and current in the opposite direction emits the other color. Alternating the two colors with sufficient frequency causes the appearance of a blended third color. For example, a red/green LED operated in this fashion will color blend to emit a yellow appearance.
- Tri-color LEDs are two LEDs in one case, but the two LEDs are connected to separate leads so that the two LEDs can be controlled independently and lit simultaneously. A three-lead arrangement is typical with one common lead (anode or cathode).[citation needed]
- RGB LEDs contain red, green and blue emitters, generally using a four-wire connection with one common lead (anode or cathode). These LEDs can have either common positive or common negative leads. Others however, have only two leads (positive and negative) and have a built in tiny electronic control unit.
- Alphanumeric LED displays are available in seven-segment and starburst format. Seven-segment displays handle all numbers and a limited set of letters. Starburst displays can display all letters. Seven-segment LED displays were in widespread use in the 1970s and 1980s, but rising use of liquid crystal displays, with their lower power needs and greater display flexibility, has reduced the popularity of numeric and alphanumeric LED displays.
Power sources
Main article: LED power sources
The current/voltage characteristic of an LED is similar to other diodes, in that the current is dependent exponentially on the voltage (see Shockley diode equation). This means that a small change in voltage can cause a large change in current. If the maximum voltage rating is exceeded by a small amount, the current rating may be exceeded by a large amount, potentially damaging or destroying the LED. The typical solution is to use constant current power supplies, or driving the LED at a voltage much below the maximum rating. Since most common power sources (batteries, mains) are not constant current sources, most LED fixtures must include a power converter. However, the I/V curve of nitride-based LEDs is quite steep above the knee and gives an If of a few milliamperes at a Vf of 3 V, making it possible to power a nitride-based LED from a 3 V battery such as a coin cell without the need for a current limiting resistor.
Electrical polarity
Main article: Electrical polarity of LEDs
As with all diodes, current flows easily from p-type to n-type material.[77] However, no current flows and no light is emitted if a small voltage is applied in the reverse direction. If the reverse voltage grows large enough to exceed the breakdown voltage, a large current flows and the LED may be damaged. If the reverse current is sufficiently limited to avoid damage, the reverse-conducting LED is a useful noise diode.
Safety
The vast majority of devices containing LEDs are "safe under all conditions of normal use", and so are classified as "Class 1 LED product"/"LED Klasse 1". At present, only a few LEDs—extremely bright LEDs that also have a tightly focused viewing angle of 8° or less—could, in theory, cause temporary blindness, and so are classified as "Class 2".[78] In general, laser safety regulations—and the "Class 1", "Class 2", etc. system—also apply to LEDs.[79]
Advantages
- Efficiency: LEDs emit more light per watt than incandescent bulbs.[80] Their efficiency is not affected by shape and size, unlike Fluorescent light bulbs or tubes.
- Color: LEDs can emit light of an intended color without using any color filters as traditional lighting methods need. This is more efficient and can lower initial costs.
- Size: LEDs can be very small (smaller than 2 mm2[81]) and are easily populated onto printed circuit boards.
- On/Off time: LEDs light up very quickly. A typical red indicator LED will achieve full brightness in under a microsecond.[82] LEDs used in communications devices can have even faster response times.
- Cycling: LEDs are ideal for uses subject to frequent on-off cycling, unlike fluorescent lamps that fail faster when cycled often, or HID lamps that require a long time before restarting.
- Dimming: LEDs can very easily be dimmed either by pulse-width modulation or lowering the forward current.
- Cool light: In contrast to most light sources, LEDs radiate very little heat in the form of IR that can cause damage to sensitive objects or fabrics. Wasted energy is dispersed as heat through the base of the LED.
- Slow failure: LEDs mostly fail by dimming over time, rather than the abrupt failure of incandescent bulbs.[83]
- Lifetime: LEDs can have a relatively long useful life. One report estimates 35,000 to 50,000 hours of useful life, though time to complete failure may be longer.[84] Fluorescent tubes typically are rated at about 10,000 to 15,000 hours, depending partly on the conditions of use, and incandescent light bulbs at 1,000–2,000 hours.
- Shock resistance: LEDs, being solid state components, are difficult to damage with external shock, unlike fluorescent and incandescent bulbs which are fragile.
- Focus: The solid package of the LED can be designed to focus its light. Incandescent and fluorescent sources often require an external reflector to collect light and direct it in a usable manner.
- Low toxicity: LEDs do not contain mercury, unlike fluorescent lamps.
- Fluorescent lamps are typically more efficient than LEDs (for lamps with the same CRI).
- High initial price: LEDs are currently more expensive, price per lumen, on an initial capital cost basis, than most conventional lighting technologies. The additional expense partially stems from the relatively low lumen output and the drive circuitry and power supplies needed.
- Temperature dependence: LED performance largely depends on the ambient temperature of the operating environment. Over-driving an LED in high ambient temperatures may result in overheating the LED package, eventually leading to device failure. Adequate heat sinking is needed to maintain long life. This is especially important in automotive, medical, and military uses where devices must operate over a wide range of temperatures, and need low failure rates.
- Voltage sensitivity: LEDs must be supplied with the voltage above the threshold and a current below the rating. This can involve series resistors or current-regulated power supplies.[85]
- Light quality: Most cool-white LEDs have spectra that differ significantly from a black body radiator like the sun or an incandescent light. The spike at 460 nm and dip at 500 nm can cause the color of objects to be perceived differently under cool-white LED illumination than sunlight or incandescent sources, due to metamerism,[86] red surfaces being rendered particularly badly by typical phosphor based cool-white LEDs. However, the color rendering properties of common fluorescent lamps are often inferior to what is now available in state-of-art white LEDs.
- Area light source: LEDs do not approximate a “point source” of light, but rather a lambertian distribution. So LEDs are difficult to apply to uses needing a spherical light field. LEDs cannot provide divergence below a few degrees. In contrast, lasers can emit beams with divergences of 0.2 degrees or less.[87]
- Blue hazard: There is a concern that blue LEDs and cool-white LEDs are now capable of exceeding safe limits of the so-called blue-light hazard as defined in eye safety specifications such as ANSI/IESNA RP-27.1–05: Recommended Practice for Photobiological Safety for Lamp and Lamp Systems.[88][89]
- Blue pollution: Because cool-white LEDs (i.e., LEDs with high color temperature) emit proportionally more blue light than conventional outdoor light sources such as high-pressure sodium vapor lamps, the strong wavelength dependence of Rayleigh scattering means that cool-white LEDs can cause more light pollution than other light sources. The International Dark-Sky Association discourages using white light sources with correlated color temperature above 3,000 K.[90]
- Droop: The efficiency of LEDs tends to decrease as one increases current.[91][92][93][94]


LED lighting in the aircraft cabin of an Airbus A320 Enhanced.


A large LED display behind a disc jockey.


LED destination displays on buses, one with a colored route number.


LED digital display that can display 4 digits along with points.


Traffic light using LED


Western Australia Police car using LED


Printhead of an Oki LED printer


LED daytime running lights of Audi A4


LED panel light source used in an experiment on plant growth. The findings of such experiments may be used to grow food in space on long duration missions.
LED uses fall into four major categories:
- Visual signals where light goes more or less directly from the source to the human eye, to convey a message or meaning.
- Illumination where light is reflected from objects to give visual response of these objects.
- Measuring and interacting with processes involving no human vision.[95]
- Narrow band light sensors where LEDs operate in a reverse-bias mode and respond to incident light, instead of emitting light.
The low energy consumption, low maintenance and small size of modern LEDs has led to uses as status indicators and displays on a variety of equipment and installations. Large area LED displays are used as stadium displays and as dynamic decorative displays. Thin, lightweight message displays are used at airports and railway stations, and as destination displays for trains, buses, trams, and ferries.
One-color light is well suited for traffic lights and signals, exit signs, emergency vehicle lighting, ships' navigation lights or lanterns (chromacity and luminance standards being set under the Convention on the International Regulations for Preventing Collisions at Sea 1972, Annex I and the CIE) and LED-based Christmas lights. In cold climates, LED traffic lights may remain snow covered.[96] Red or yellow LEDs are used in indicator and alphanumeric displays in environments where night vision must be retained: aircraft cockpits, submarine and ship bridges, astronomy observatories, and in the field, e.g. night time animal watching and military field use.
Because of their long life and fast switching times, LEDs have been used in brake lights for cars high-mounted brake lights, trucks, and buses, and in turn signals for some time, but many vehicles now use LEDs for their rear light clusters. The use in brakes improves safety, due to a great reduction in the time needed to light fully, or faster rise time, up to 0.5 second faster than an incandescent bulb. This gives drivers behind more time to react. It is reported that at normal highway speeds, this equals one car length equivalent in increased time to react. In a dual intensity circuit (i.e., rear markers and brakes) if the LEDs are not pulsed at a fast enough frequency, they can create a phantom array, where ghost images of the LED will appear if the eyes quickly scan across the array. White LED headlamps are starting to be used. Using LEDs has styling advantages because LEDs can form much thinner lights than incandescent lamps with parabolic reflectors.
Due to the relative cheapness of low output LEDs, they are also used in many temporary uses such as glowsticks, throwies, and the photonic textile Lumalive. Artists have also used LEDs for LED art.
Weather/all-hazards radio receivers with Specific Area Message Encoding (SAME) have three LEDs: red for warnings, orange for watches, and yellow for advisories & statements whenever issued.
Lighting
Main article: LED lamp
With the development of high efficiency and high power LEDs it has grown possible to use LEDs in lighting and illumination. Replacement light bulbs have been made, as well as dedicated fixtures and LED lamps. LEDs are used as street lights and in other architectural lighting where color changing is used. The mechanical robustness and long lifetime is used in automotive lighting on cars, motorcycles and on bicycle lights.
LED street lights are employed on poles and in parking garages. In 2007, the Italian village Torraca was the first place to convert its entire illumination system to LEDs.[97]
LEDs are used in aviation lighting. Airbus has used LED lighting in their Airbus A320 Enhanced since 2007, and Boeing plans its use in the 787. LEDs are also being used now in airport and heliport lighting. LED airport fixtures currently include medium intensity runway lights, runway centerline lights and obstruction lighting.
LEDs are also suitable for backlighting for LCD televisions and lightweight laptop displays and light source for DLP projectors (See LED TV). RGB LEDs raise the color gamut by as much as 45%. Screens for TV and computer displays can be made thinner using LEDs for backlighting.[98]
LEDs are used increasingly commonly in aquarium lights. Particularly for reef aquariums, LED lights provide an efficient light source with less heat output to help maintain optimal aquarium temperatures. LED-based aquarium fixtures also have the advantage of being manually adjustable to emit a specific color-spectrum for ideal coloration of corals, fish, and invertebrates while optimizing photosynthetically active radiation (PAR) which raises growth and sustainability of photosynthetic life such as corals, anemones, clams, and macroalgae. These fixtures can be electronically programmed to simulate various lighting conditions throughout the day, reflecting phases of the sun and moon for a dynamic reef experience. LED fixtures typically cost up to five times as much as similarly rated fluorescent or high-intensity discharge lighting designed for reef aquariums and are not as high output to date.
The lack of IR/heat radiation makes LEDs ideal for stage lights using banks of RGB LEDs that can easily change color and decrease heating from traditional stage lighting, as well as medical lighting where IR-radiation can be harmful.
LEDs are small, durable and need little power, so they are used in hand held devices such as flashlights. LED strobe lights or camera flashes operate at a safe, low voltage, instead of the 250+ volts commonly found in xenon flashlamp-based lighting. This is especially useful in cameras on mobile phones, where space is at a premium and bulky voltage-raising circuitry is undesirable. LEDs are used for infrared illumination in night vision uses including security cameras. A ring of LEDs around a video camera, aimed forward into a retroreflective background, allows chroma keying in video productions.
LEDs are used for decorative lighting as well. Uses include but are not limited to indoor/outdoor decor, limousines, cargo trailers, conversion vans, cruise ships, RVs, boats, automobiles, and utility trucks. Decorative LED lighting can also come in the form of lighted company signage and step and aisle lighting in theaters and auditoriums.
Smart lighting
Light can be used to transmit broadband data, which is already implemented in IrDA standards using infrared LEDs. Because LEDs can cycle on and off millions of times per second, they can be wireless transmitters and access points for data transport.[99] Lasers can also be modulated in this manner.
Sustainable lighting
Efficient lighting is needed for sustainable architecture. A 13 watt LED lamp emits 450 to 650 lumens.[100] which is equivalent to a standard 40 watt incandescent bulb. [101] A standard 40 W incandescent bulb has an expected lifespan of 1,000 hours while an LED can continue to operate with reduced efficiency for more than 50,000 hours, 50 times longer than the incandescent bulb.
Environmentally friendly options
One kilowatt-hour of electricity will cause 1.34 pounds (610 g) of CO2 emission.[102] Assuming the average light bulb is on for 10 hours a day, one 40-watt incandescent bulb will cause 196 pounds (89 kg) of CO2 emission per year. The 13-watt LED equivalent will only cause 63 pounds (29 kg) of CO2 over the same time span. A building’s carbon footprint from lighting can be reduced by 68% by exchanging all incandescent bulbs for new LEDs in warm climates. In cold climates, the energy saving may be lower, since more heating is needed to compensate for the lower temperature.
LEDs are also non-toxic unlike the more popular energy efficient bulb option: the compact fluorescent a.k.a. CFL which contains traces of harmful mercury. While the amount of mercury in a CFL is small, introducing less into the environment is preferable.
Economically sustainable
LED light bulbs could be a cost-effective option for lighting a home or office space because of their very long lifetimes. Consumer use of LEDs as a replacement for conventional lighting system is currently hampered by the high cost and low efficiency of available products. 2009 DOE testing results showed an average efficacy of 35 lm/W, below that of typical CFLs, and as low as 9 lm/W, worse than standard incandescents.[100] The high initial cost of the commercial LED bulb is due to the expensive sapphire substrate which is key to the production process. The sapphire apparatus must be coupled with a mirror-like collector to reflect light that would otherwise be wasted.
In 2008, a materials science research team at Purdue University succeeded in making LED bulbs with a substitute for the sapphire components.[103] The team used metal-coated silicon wafers with a built-in reflective layer of zirconium nitride to lessen the overall production cost of the LED. They predict that within a few years, LEDs produced with their revolutionary new method will be competitively priced with CFLs. The less expensive LED would not only be the best energy saver, but also a low cost bulb.
Non-visual applications
Light has many other uses besides for seeing. LEDs are used for some of these. The uses fall in three groups: Communication, sensors and light matter interaction.
The light from LEDs can be modulated very quickly so they are used extensively in optical fiber and Free Space Optics communications. This include remote controls, such as for TVs and VCRs, where infrared LEDs are often used. Opto-isolators use an LED combined with a photodiode or phototransistor to provide a signal path with electrical isolation between two circuits. This is especially useful in medical equipment where the signals from a low voltage sensor circuit (usually battery powered) in contact with a living organism must be electrically isolated from any possible electrical failure in a recording or monitoring device operating at potentially dangerous voltages. An optoisolator also allows information to be transferred between circuits not sharing a common ground potential.
Many sensor systems rely on light as the signal source. LEDs are often ideal as a light source due to the requirements of the sensors. LEDs are used as movement sensors, for example in optical computer mice. The Nintendo Wii's sensor bar uses infrared LEDs. In pulse oximeters for measuring oxygen saturation. Some flatbed scanners use arrays of RGB LEDs rather than the typical cold-cathode fluorescent lamp as the light source. Having independent control of three illuminated colors allows the scanner to calibrate itself for more accurate color balance, and there is no need for warm-up. Further, its sensors only need be monochromatic, since at any one time the page being scanned is only lit by one color of light. Touch sensing: Since LEDs can also be used as photodiodes, they can be used for both photo emission and detection. This could be used in for example a touch-sensing screen that register reflected light from a finger or stylus.[104]
Many materials and biological systems are sensitive to, or dependent on light. Grow lights use LEDs to increase photosynthesis in plants[105] and bacteria and viruses can be removed from water and other substances using UV LEDs for sterilization.[57] Other uses are as UV curing devices for some ink and coating methods, and in LED printers.
Plant growers are interested in LEDs because they are more energy efficient, emit less heat (can damage plants close to hot lamps), and can provide the optimum light frequency for plant growth and bloom periods compared to currently used grow lights: HPS (high pressure sodium), MH (metal halide) or CFL/low-energy. However, LEDs have not replaced these grow lights due to higher price. As mass production and LED kits develop, the LED products will become cheaper.
LEDs have also been used as a medium quality voltage reference in electronic circuits. The forward voltage drop (e.g., about 1.7 V for a normal red LED) can be used instead of a Zener diode in low-voltage regulators. Red LEDs have the flattest I/V curve above the knee. Nitride-based LEDs have a fairly steep I/V curve and are useless for this purpose. Although LED forward voltage is far more current-dependent than a good Zener, Zener diodes are not widely available below voltages of about 3 V.
Light sources for machine vision systems
Machine vision systems often require bright and homogeneous illumination, so features of interest are easier to process. LEDs are often used for this purpose, and this is likely to remain one of their major uses until price drops low enough to make signaling and illumination uses more widespread. Barcode scanners are the most common example of machine vision, and many low cost ones use red LEDs instead of lasers. Optical computer mice are also another example of LEDs in machine vision, as it is used to provide an even light source on the surface for the miniature camera within the mouse. LEDs constitute a nearly ideal light source for machine vision systems for several reasons:
The size of the illuminated field is usually comparatively small and machine vision systems are often quite expensive, so the cost of the light source is usually a minor concern. However, it might not be easy to replace a broken light source placed within complex machinery, and here the long service life of LEDs is a benefit.
LED elements tend to be small and can be placed with high density over flat or even shaped substrates (PCBs etc.) so that bright and homogeneous sources can be designed which direct light from tightly controlled directions on inspected parts. This can often be obtained with small, low cost lenses and diffusers, helping to achieve high light densities with control over lighting levels and homogeneity. LED sources can be shaped in several configurations (spot lights for reflective illumination; ring lights for coaxial illumination; back lights for contour illumination; linear assemblies; flat, large format panels; dome sources for diffused, omnidirectional illumination).
LEDs can be easily strobed (in the microsecond range and below) and synchronized with imaging. High power LEDs are available allowing well lit images even with very short light pulses. This is often used to obtain crisp and sharp “still” images of quickly moving parts.
LEDs come in several different colors and wavelengths, allowing easy use of the best color for each need, where different color may provide better visibility of features of interest. Having a precisely known spectrum allows tightly matched filters to be used to separate informative bandwidth or to reduce disturbing effects of ambient light. LEDs usually operate at comparatively low working temperatures, simplifying heat management and dissipation. This allows using plastic lenses, filters, and diffusers. Waterproof units can also easily be designed, allowing use in harsh or wet environments (food, beverage, oil industries).


Comparison of LCD, edge lit LED and LED TVs
A LED-backlight LCD television is an LCD TV that uses LED backlighting[1][2] rather than the cold cathode fluorescent lights (CCFLs) used in traditional LCD televisions. It is not a true LED display but is often called "LED TV" by some manufacturers.[3] The use of LED backlighting has a dramatic impact, resulting in a thinner panel, less power consumption, and a brighter display with better contrast levels. It also generates less heat than an ordinary LCD TV.
The LEDs can come in three forms: dynamic RGB LEDs which are positioned behind the panel, white Edge-LEDs positioned around the rim of the screen which use a special diffusion panel to spread the light evenly behind the screen (the most common) and full-array which are arranged behind the screen but they are incapable of dimming or brightening individually.[4][5]
LED backlighting techniques
RGB dynamic LEDs
This method of backlighting allows dimming to occur in locally specific areas of darkness on the screen. This can show truer blacks, whites and PRs[clarification needed] at much higher dynamic contrast ratios, at the cost of less detail in small bright objects on a dark background, such as star fields.[6]
Edge-LEDs
This method of backlighting allows for LED-backlit TVs to become extremely thin. The light is diffused across the screen by a special panel which produces a uniform color range across the screen.
Full Array LEDs
Many brands use LED backlighting technology and may offer a range of benefits over CCFL LCD TVs such as reduced energy consumption, better contrast and brightness, greater colour range, more rapid response to changes in scene and a capacity to provide the means to render an image more accurately.[7]
Differences between LED-backlit and CCFL-backlit LCD displays
LED-backlit LCD TVs differ from conventional CCFL-backlit LCD TVs in the following:
- Produce images with greater dynamic contrast.[8]
- With Edge-LED lighting they can be extremely slim. Models on the market can be approximately one inch thick.[8]
- Offer a wider color gamut, especially when RGB-LED backlighting is used.[9]
- Less environmental pollution on disposal.[8]
- Higher sales price.[8]
- Generally 20-30% lower power consumption.[citation needed]
TV manufacturers can use an LED backlight instead of the standard Cold Cathode Fluorescent Lamps (LCD-CCFL) used in most LCD televisions. It is important to distinguish this method of simply backlighting a conventional LCD panel, from a hypothetical true LED display, or an Organic light-emitting diode (OLED) display. LCD-based televisions described as 'LED TVs' are vastly different from self-illuminating OLED, OEL or AMOLED display technologies. In terms of the use of the term 'LED TV' in the UK, the ASA (Advertising Standards Authority) has made it clear in prior correspondence that it does not object to the use of the term, but does require it to be clarified in any advertising. There are several methods of backlighting an LCD panel using LEDs including the use of either White or RGB (Red, Green and Blue) LED arrays positioned behind the panel; and Edge-LED lighting, which uses white LEDs arranged around the inside frame of the TV along with a special light diffusion panel designed to spread the light evenly behind the LCD panel.
An LED backlight offers several general benefits over regular CCFL backlight TVs, typically higher brightness. Compared to regular CCFL backlighting, there may also be benefits to color gamut. However advancements in CCFL technology mean wide color gamuts and lower power consumption are also possible. The principal barrier to wide use of LED backlighting on LCD televisions is cost.
The variations of LED backlighting do offer different benefits. The first commercial LED backlit LCD TV was the Sony Qualia 005 (introduced in 2004). This featured RGB LED arrays to offer a color gamut around twice that of a conventional CCFL LCD television (the combined light output from red, green and blue LEDs produces a more pure white light than is possible with a single white light LED). RGB LED technology continues to be used on selected Sony BRAVIA LCD models, with the addition of 'local dimming' which enables excellent on-screen contrast through selectively turning off the LEDs behind dark parts of a picture frame.
Edge LED lighting was also first introduced by Sony (September 2008) on the 40 inch BRAVIA KLV-40ZX1M (referred to as the ZX1 in Europe). The principal benefit of Edge-LED lighting for LCD televisions is the ability to build thinner housings (the BRAVIA KLV-40ZX1M is as thin as 9.9mm). Samsung has also introduced a range of Edge-LED lit LCD televisions with extremely thin housings.
LED-backlit LCD TVs are considered a more sustainable choice, with a longer life and better energy efficiency than plasmas and conventional LCD TVs.[10] Unlike CCFL backlights, LEDs also use no mercury in their manufacture. However, other elements such as gallium and arsenic are used in the manufacture of the LED emitters themselves, meaning there is some debate over whether they are a significantly better long term solution to the problem of TV disposal.
Because LEDs are able to be switched on and off more quickly than CCFL displays and can offer a higher light output, it is theoretically possible to offer very high contrast ratios. They can produce deep blacks (LEDs off) and a high brightness (LEDs on), however care should be taken with measurements made from pure black and pure white outputs, as technologies like Edge-LED lighting do not allow these outputs to be reproduced simultaneously on-screen.
In September 2009 Nanoco Group announced that it has signed a joint development agreement with a major Japanese electronics company under which it will design and develop quantum dots for LED Backlights in LCD televisions.[11] Quantum dots are valued for displays, because they emit light in very specific gaussian distributions. This can result in a display that more accurately renders the colors than the human eye can perceive. Quantum dots also require very little power since they are not color filtered. In September 2010, LG Electronics revealed their new product which claimed as the world's slimmest full LED 3D TV at the IFA consumer electronics trade show in Berlin.[12]






